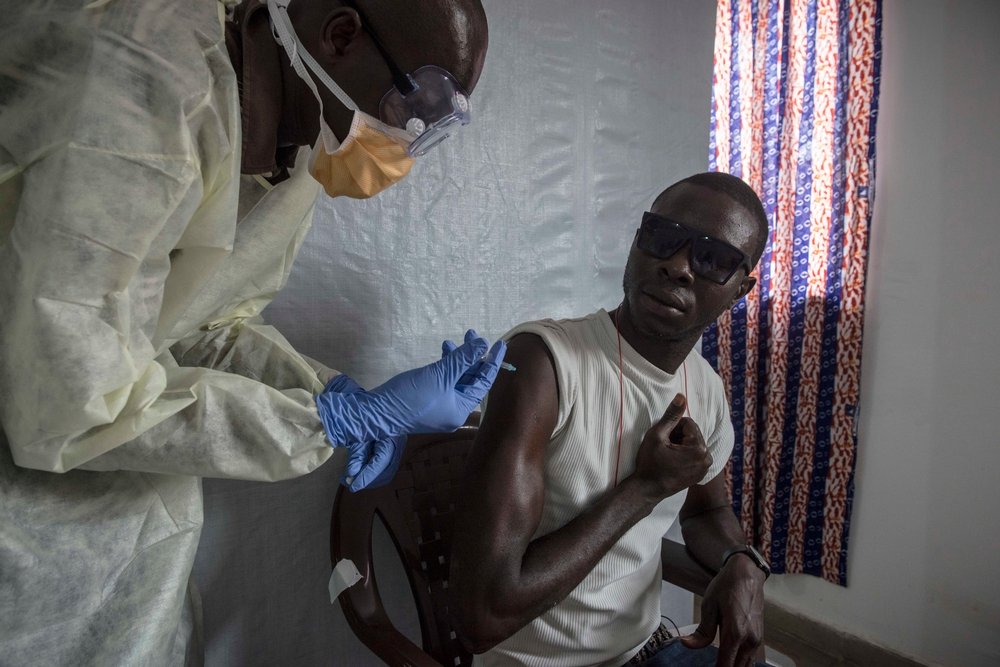
Massive partnership works in record time to test vaccine and show efficacy and safety
Results of tests of a new vaccine to protect against Ebola indicate a high level of efficacy in a trial involving more than 4,000 persons in close contact with the deadly virus in Guinea, one of the three West African countries stricken by Ebola.
The Lancet, the prestigious British medical journal, published the results of the trials on-line on July 31.
The trial results indicate that the new vaccine called VSV-ZEBOV is 100 percent efficacious in about 10 days after being administered to a person without the infection.
"Having seen the devastating effects of Ebola on communities and even whole countries with my own eyes, I am very encouraged by today's news," says Børge Brende, Minister of Foreign Affairs in Norway.
"This new vaccine, if the results hold up, may be the silver bullet against Ebola, helping to bring the current outbreak to zero and to control future outbreaks of this kind. I would like to thank all partners who have contributed to achieve this sensational result, due to an extra-ordinary and rapid collaborative effort."
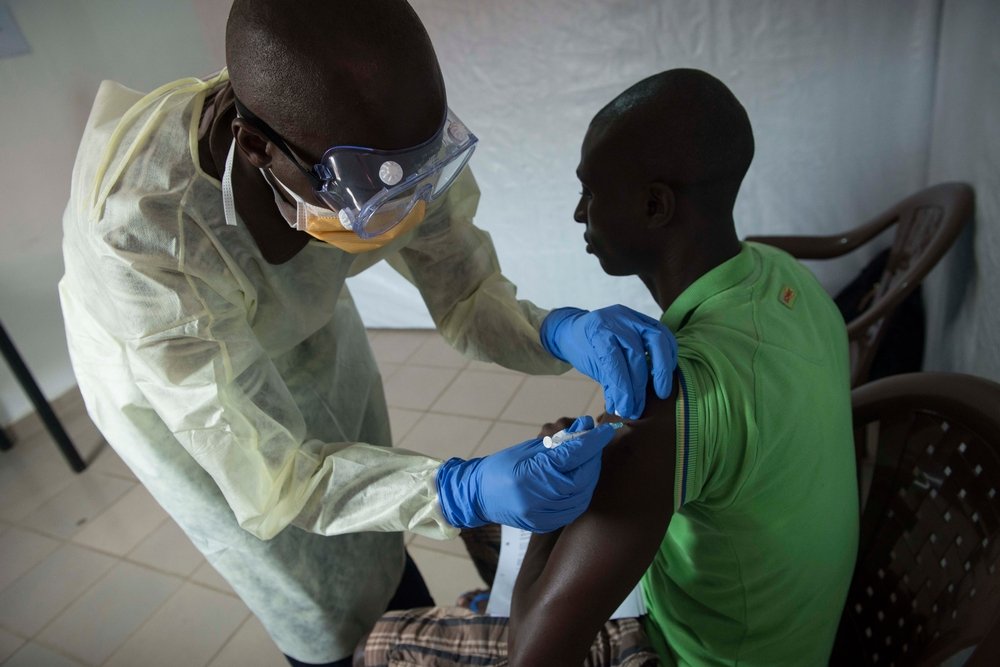
Because of the urgency of the Ebola crisis, clinical development of this vaccine proceeded from initial testing in humans to demonstration of vaccine efficacy and safety in a large Phase III study in Guinea in less than 12 months, a record time.
"Normally, it takes about a decade or more for a vaccine to come to this point," says Professor Peter Smith, D.Sc., an epidemiologist and specialist in vaccine trials, from the London School of Hygiene & Tropical Medicine and a key advisor.
Because of the Ebola crisis, a massive team was assembled which included scientists, physicians, epidemiologists and experts from the World Health Organization (WHO), Norway, Canada, Guinea, Doctors without Borders, Universities of Florida, Maryland and Bern, and the London School of Hygiene & Tropical Medicine.
"We knew it was a race against time and that the trial had to be implemented under the most challenging circumstances," says John-Arne Røttingen, M.D., PhD., head of infectious disease control at the Norwegian Institute of Public Health and chair of the trial's steering group.
Funding came from the Wellcome Trust, Norway, Canada, WHO, Doctors without Borders. Merck (known as MSD outside the United States and Canada), one of the world's leading vaccines and pharmaceutical companies, based in Kenilworth, NJ., provided the vaccine. Scientists from the company, along with those from NewLink Genetics Corporation, an early developer of the vaccine, also gave detailed technical support to field trial staff on the vaccine and its administration.
"Health workers have been fighting an unfair battle against the Ebola and for the first time there is a prospect of a tool that could protect lives and break chains of transmission," says Bertrand Draguez, M.D, medical director of Doctors without Borders, the first international organization to treat and follow up Ebola patients in Guinea.
"New technologies are critically needed to diagnose, treat and prevent Ebola and other emerging diseases".
"We view this promising vaccine candidate as a global resource in the fight against Ebola," says Gregory Taylor, M.D., Canada's Chief Public Health Officer.
"It is very encouraging to see the vaccine, which is the product of many years of research and innovation at the Public Health Agency of Canada's National Microbiology Laboratory, receive positive results in this clinical trial. Having a regulated, safe and effective vaccine that has undergone clinical trials will be an important tool in further containing the current outbreak and providing protection against possible future outbreaks."
The Public Health Agency of Canada, which maintains ownership of the intellectual property associated with VSV-EBOV, did the initial research and development on the vaccine. Merck, the pharmaceutical giant with its massive vaccine program, entered into an exclusive license agreement in November 2014, and assumed responsibility for efforts to research, development, manufacture and distribute the vaccine which was tested in the trial.
"In Canada, we have significant expertise in vaccine research and have built the capacity to rapidly respond to global health threats like the Ebola outbreak," says Dr. Jane Aubin, Chief Scientific Officer and Vice-President, Research and Knowledge Translation, Canadian Institutes of Health Research (CIHR). "It's encouraging to see the positive results of this trial on a Canadian-made vaccine."
CIHR funded a clinical trial of the vaccine in Halifax, Canada, in partnership with the Public Health Agency of Canada. These two agencies also collaborated with Canada's International Development Research Centre and the Department of Foreign Affairs, Trade and Development Canada, to provide funding for, and expert guidance on, the trial in Guinea.
"Canada's International Development Research Centre, the Public Health Agency of Canada, and our Department of Foreign Affairs, Trade and Development ensured the safety of the trial in the field," says IDRC president Jean Lebel. "We did this ground level work with local Guinean partners, creating buy-in for the trial and building their clinical research capacity for the future. Our shared goal, among all partners, is ultimately saving lives. This is about more than a vaccine or statistics. This is about real people, who are parents, children, siblings, community members."
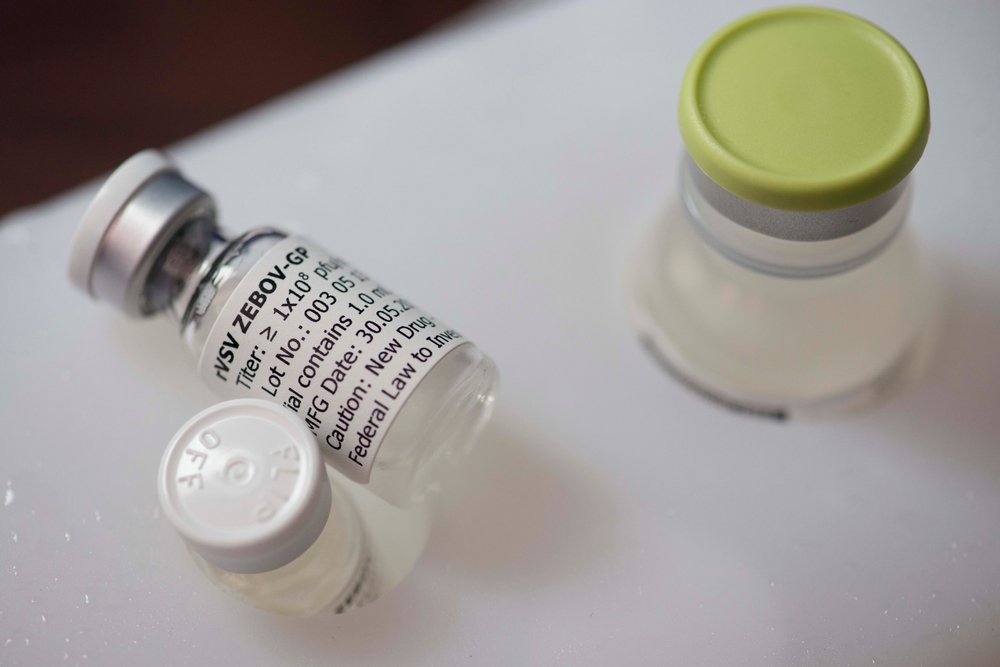
The vaccine
The Public Health Agency of Canada and NewLink Genetics Corporation did the initial research on the vaccine. Merck licensed the vaccine in November 2014 and assumed responsibility for efforts to research, develop, manufacture and distribute the vaccine, which was tested in the trial.
The vaccine combines a gene encoding a key protein of the Zaire strain of the Ebola virus, which ravaged West Africa, and another unrelated virus -VSV. Combination of these components results in a weakened vaccine virus that cannot cause disease, but that does stimulate the body to generate an immune response. In this way, the vaccine triggers the production of antibodies to fight off the disease and is made safe.
"The VSV-ZEBOV vaccine works in a similar way to live-attenuated vaccines against other viral infections," says Mark Feinberg, M.D, Ph.D., of Merck Vaccines. "The ability of this vaccine to elicit prompt immune responses against theEbola virus following administration of a single dose represents an important attribute."
After VSV-ZEBOV is further studied and licensed by regulatory authorities, Merck plans to produce enough doses to control future outbreaks. "It will be stockpiled for other Ebola emergencies," says Dr. Feinberg.
At this point, the vaccine will not be used like common vaccines such as those for polio and measles, which are routinely administered.
"Ebola is not a common infectious disease, and outbreaks arise in sporadic and unpredictable ways," says Dr. Feinberg. "So the vaccine will likely be used in at-risk communities where and when it is required. However, it will also hopefully provide an important additional approach to help protect health workers who care for Ebola-infected patients."
"We are doing additional research on the vaccine to provide a comprehensive set of data to inform licensure decisions by regulatory agencies and to facilitate the development and implementation of policy decisions by key international and national authorities," says Dr. Feinberg.
A drawback to the current formulation of the vaccine is that it must be kept very cold in a tropical country with sparse electricity. Future work will be needed to develop a more thermostable formulation.
The outbreak in Liberia, Sierra Leone and Guinea was caused by the Ebola strain called Zaire. Other Ebola virus strains that have been seen in previous outbreaks, such as Sudan, may require the development of new related vaccines. But the development of such new vaccines has been hugely helped by this study.
The trial
"This trial dared to use a highly innovative and pragmatic design, which allowed the team in Guinea to assess this vaccine in the middle of an epidemic," says Professor Jeremy Farrar, M.D., Ph.D., director of the Wellcome Trust and one of the world's leading experts on infectious disease. "It is a remarkable result which shows the power of equitable international partnerships and flexibility."
"Our hope is that this vaccine will now help bring this epidemic to an end and be available for the inevitable future Ebola epidemics. This partnership also shows that such critical work is possible in the midst of a terrible epidemic. It should change how the world responds to such emerging infectious disease threats."
The vaccine had been tested on animals successfully, but not initially on humans. Thus, it was unknown if the vaccine would be well tolerated and stimulate a positive immune response in humans and thereby protect individuals from infection.
"In early human trials, the vaccine had only transitory side effects -such as fever and joint pain- in a few of the individuals who received the vaccine," says Professor Smith. "These side effects were generally mild and of a limited duration and are consistent with those seen with a number of other live viral vaccines."
In a recent review of the available safety data, the WHO's Global Advisory Committee on Vaccine Safety concluded that the VSV-ZEBOV vaccine has an acceptable safety profile. In addition to the study in Guinea, the vaccine is being studied in large clinical trials conducted by the U.S. National Institutes of Health and their partners in Liberia, and by the U.S. Center of Disease Control and Prevention and their partners in Sierra Leone.
In September 2014, world-leading experts assembled at the WHO in Geneva, Switzerland. At that meeting, it was decided to prioritize clinical testing of two vaccine candidates. In early October, Norway decided to support vaccine trials in Guinea. On the margins of a high-level vaccine meeting at WHO on October 23rd, a working group for vaccine trials in Guinea was established and a November 5th meeting of this group decided to move forward with the trial based on financial commitments from Norway.
The design of the trial was unique in a number of ways. Firstly, the experts used a ring vaccination strategy, developed to control smallpox from the mid-1960s to its eradication in 1980. It isolated smallpox patients by vaccinating family members and other close contacts, creating a ring barrier around the disease.
Using the ring approach, when the Ebola epidemic in Guinea had dispersed into smaller local outbreaks, the vaccinators and the trial team were able to move with it. It allowed the trial to continue and at the same time contribute to the control the Ebola outbreak seamlessly.
The volunteers who received the vaccine came from areas in Guinea that had Ebola outbreaks. These volunteers were directly in contact with those who had contracted the virus or were contacts-of-contacts, such as neighbors, classmates or extended family.
In some cases, the clusters were whole villages. In others, the clusters were smaller sections of towns and cities.
Secondly, the experts abandoned the placebo régime used in many clinical trials, because of ethical concerns. The volunteers were still randomized into two groups. The intervention group received the vaccine immediately. The control group received the vaccine three weeks later. This replaced the classical placebo régime, but the trial is still a randomized controlled trial.
The results found that within 10 days the vaccine protected against the Ebola virus for both groups.
"The global health community may now have a very important new tool to beat Ebola", says Dr. Røttingen. "This trial has showed us how to isolate this virus with a vaccine and protect entire villages. We are doing that through a public health intervention like the ring vaccination strategy."
"More importantly, it showed us the power of a well-organized international partnership working hand-in-hand to overcome a public health emergency in record time."
"We have new confidence from the lessons we have learned with the development of the Ebola vaccine and the trial that so far indicates remarkable efficacy. Inevitably, further analyses and a longer follow up are needed but this is a wonderful result."
"These encouraging results from the Ebola vaccine trials show how WHO, working with partners and the international community, was able to advance rapidly on Ebola research and development," says Margaret Chan, M.D., Director-General of WHO.
"By compressing into months a process that usually takes years, we have developed potential new tools in our continuing battle against Ebola. The governments and all those involved in the vaccine trials deserve our thanks and backing."
Find out more about MSF in Guinea.